 It's a polymorphism that we could use to follow the inheritance of DNA. That sequence difference doesn't necessarily mean that there's a disease associated with it. So these differences in nucleic acid sequences and restriction enzyme binding sites just mean that there's a difference in the sequence between those two people. In one person, without the enzyme site you'll see one band, and the person that has the enzyme site, you'll see two bands, representing the two cleaved products. We typically see these, or we monitor these, by isolating the DNA, cutting it with that bacterial restriction enzyme, and running it on a gel using electrophoresis. The ligated vector and DNA fragment can now be transformed into a host cell for replication and expression. The enzyme DNA ligase is used to seal together restriction fragments by forming new phosphodiester bonds. To join DNA molecules together, an enzyme called DNA ligase is used. And that results in a polymorphism, or difference between those two people. Recall that restriction endonucleases are used to cut DNA. So then, if you isolate that piece of DNA surrounding that site from two people, from one of them it will be cut by the enzyme and the other one it won't. Genome Editing Reduce Star Activity with High-Fidelity Restriction Enzymes. So a single base difference between two people could result in either the presence or absence of that restriction site. fragments, PCR cloning, and site-directed mutagenesis. So why is that useful? Well, we can take advantage of this fact to actually look for differences between people if they have that restriction enzyme site or not. What is it, though? So basically, if you follow the sequence of DNA, particular sites, a series of four to eight nucleic acids, results in a restriction site where an enzyme from bacteria can actually bind and cleave that DNA. Specifically, we currently work on (1) mitochondrial dynamics and genome instability, (2) arginine and tumor progression and (3) autophagy and drug resistance.RFLPs have been very useful to use as markers for following a genomic DNA, either from human or other animals. Ann’s team is to decipher the signaling transduction pathways and molecular mechanisms underlying adaptive response to metabolic stress. respectively, we have identified eight polymorphic restriction sites.  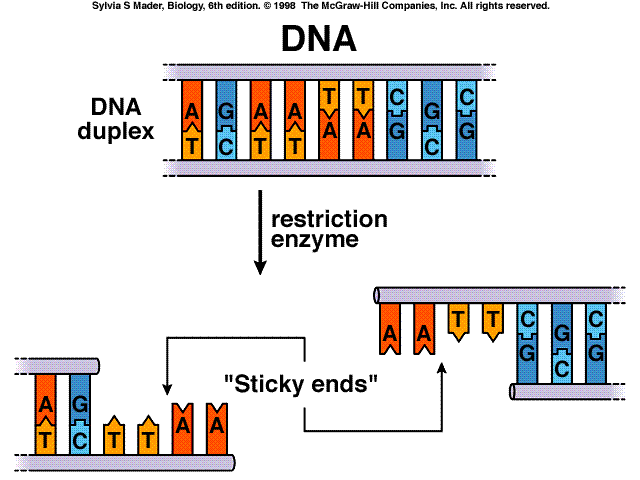
Their results define a mechanism of mitochondrial dynamics regulation that operates in cancer cells under metabolic stress to help preserve their survival, with potential therapeutic implications. DNA restriction fragments associated with 1-antitrypsin indicate a single origin. Very recently, they found that metabolic stress by restriction of arginine or glucose or inhibition of fatty acid oxidation leads to KAP1 Ser473 phosphorylation to regulate mitochondrial fusion and fission. Their data suggest that arginine depletion results in a decrease of mitochondrial inner membrane and matrix proteins that impairs mitochondrial oxidative phosphorylation (OxPhos) and respiratory function, so-called metabolic stress. While it has long been recognized that some cancer cells are resistant to apoptosis, they discovered an alternative pathway to induce cell death that overcomes resistance to traditional apoptosis associated with cancer. Unlike apoptosis, a cell-death process in which DNA is damaged within the cell nucleus, in chromatin autophagy the nucleus is fragmented and its pieces shuttled off to the lysosome (an organelle within the cell membrane) where the fragments are degraded. Their results describe how arginine starvation specifically kills tumor cells by a novel mechanism involving mitochondria dysfunction, generation of reactive oxygen species, nuclear DNA leakage and chromatophagy, where leaked DNA is captured and “eaten” by giant autophagosomes. The enzyme cuts the double-stranded DNA, resulting in DNA fragments. His research team focuses on therapeutic implication of ASS1 (argininosuccinate synthetase 1) in breast cancer biology, and we made several novel discoveries by providing a mechanistic link between ASS1 deficiency, arginine deprivation and lethal chromatin autophagy.įurthermore, they identify that arginine deprivation as a new potential breast and prostate cancer therapy that targets a subtype of these cancers that has low ASS1 expression. Each restriction enzyme moves along a DNA molecule until it finds a specific recognition sequence in the DNA. David Ann, Ph.D., is interested in the role of post-translational modifications including phosphorylation, acetylation, sumoylation, O-glycosylation and ubiquitination in human cancers, with a particular focus on oncogenesis, metastasis and cancer metabolism. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
